Saisei Medical Conference 2025
📍 Osaka International Convention Centre, 📅 Saturday, November 8, 2025 | 10:00 AM – 5:00 PM A Global Gathering in Osaka This November, Saisei Mirai Clinic proudly presents the Saisei Medical…
What Is MAF? A Simple Guide to Immune Balance and Healthy Aging
When people begin exploring immune balance, healthy aging, immune resilience, and long-term wellness, they often come across the term MAF. But what does MAF actually mean? And why is it…
Link to Age-Related Diseases
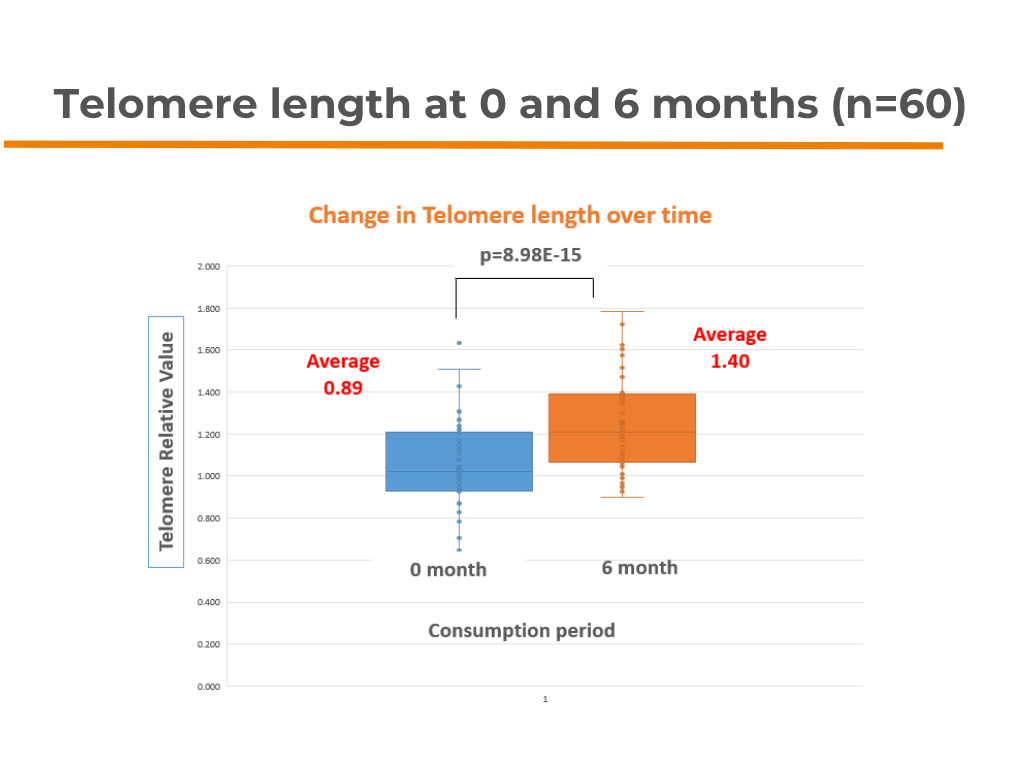
Shortened telomeres have been associated with age-related conditions such as cardiovascular diseases, neurodegenerative disorders, and weakened immune function.
Age well, beautifully and intelligently
MAF Capsules Triple is a refined, food-based daily format designed for adults who want to support longevity, vitality and healthy aging in a more thoughtful, consistent way.
General recommended use is 1–3 capsules daily on an empty stomach with water.

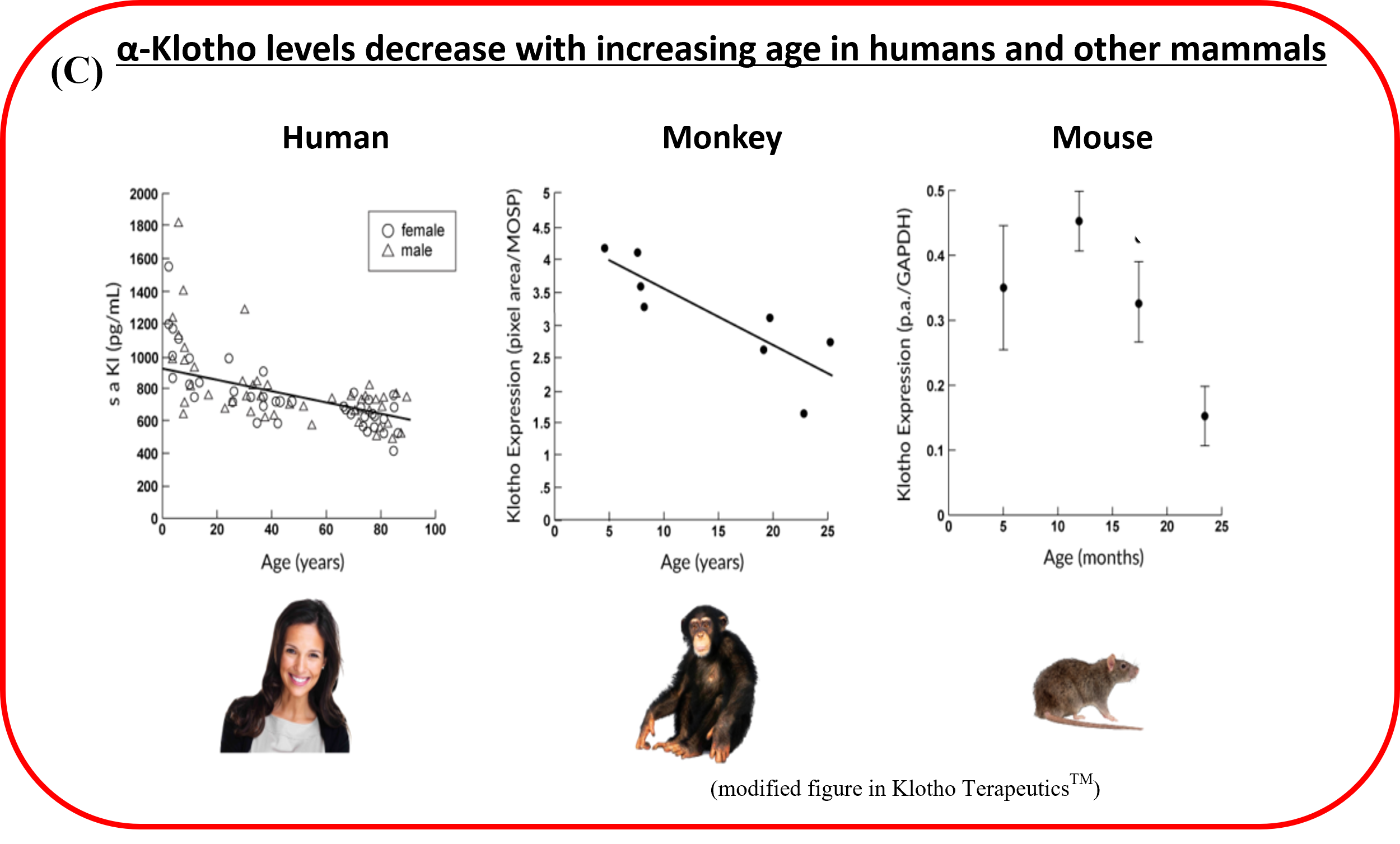
Support Healthy Aging Through Telomere & Klotho Pathways
Aging begins at the cellular level. Telomere shortening, declining Klotho expression, and immune imbalance are all linked to how the body changes over time.
Aging often begins subtly, long before it feels obvious
As we age, longevity-related factors such as α-Klotho naturally decline. For adults focused on beauty, vitality and healthy aging, this stage of life often becomes the point where consistency matters more.
Wellness After 40
A 60-second story for life after 40.
In our 40s, many of us begin to notice changes in energy, focus, and resilience. As we age, Klotho levels may decline and telomeres naturally shorten over time. This short comic explains why daily support from within may matter.
Support Healthy Aging From Within
MAF Triple is a Japan-developed wellness supplement designed to support immune balance, cellular wellness, and long-term vitality as part of your healthy-aging routine.
Explore MAF TripleThis product is a wellness supplement intended to support general health. It is not intended to diagnose, treat, cure, or prevent any disease.
Telomeres & α-Klotho: Two Important Pathways in Healthy Aging
Telomeres and α-Klotho are both closely connected to cellular aging, resilience, and long-term vitality. As we age, these pathways naturally change, which is why maintaining cellular balance becomes increasingly important after 40.

Telomeres
Cellular aging support

α-Klotho
Longevity-related factor
Why this matters after 40
Telomeres and α-Klotho are both closely linked to how the body ages at a cellular level.
Supporting these pathways may be especially important for adults who want to maintain long-term vitality, beauty, and healthy aging.

Developed within the Saisei Mirai Group in Japan
MAF research is developed within the Saisei Mirai Group in Japan, where doctors and researchers explore the relationship between immune balance, cellular health and long-term aging.
Led by Dr. Toshio Inui and the wider Japanese medical team, this work reflects a thoughtful approach to healthy aging — not a quick fix, but something designed to become part of everyday life.
Saisei Australia brings this Japanese research and product philosophy to Australia and New Zealand, with local support and distribution.
Research-led, presented with care
We keep the homepage summary visual and easy to understand, while the full detail lives in the research pages.
Phase 2 study summary presented on the site.
Clear and easy for visitors to understand.
Shown at 6 months in the study summary.
Shown at 3 and 6 months in the site summary.
General recommended use: 1–3 capsules daily on an empty stomach with water.
Study note: in the Phase 2 study featured on this site, participants took 2 capsules daily.
Featured on Nature.com
Scientific topics connected to nutrition, brain health, immune function, and healthy longevity.

Exploring Links Between Nutrition & Longevity
Nutrition, immune function, healthy ageing and longevity research.
Read Article
Exploring Healthy Brain Ageing
Brain ageing, cognition, neuroinflammation and healthy brain resilience.
Read ArticleOur flagship format for people who want one strong starting point
MAF Capsules Triple is designed as the premium lead format in the Dietary MAF range, offering a clear next step for customers focused on longevity, vitality and healthy aging.

Discover more from Saisei Australia
While longevity and healthy aging remain the main focus, you can also explore broader themes related to brain health, immune balance and autism perspectives.

Brain Health
For those interested in cognitive clarity, healthy brain aging, and research on neuro-supportive pathways.
Immune Balance
Explore macrophage activation, immune resilience, and broader immune-related wellness themes.

Autism Perspective
Learn more about Saisei’s broader perspective on immune regulation, brain development and supportive research themes.
Choose the format that fits your lifestyle
Start with MAF Capsules Triple as our flagship option, or explore Capsules, Powder, and Lollies depending on the format that feels easiest to continue each day.

MAF Capsules Triple
Our strongest premium option for adults who want the lead format first.

MAF Capsules
A more approachable capsule option for those who prefer to begin with a smaller daily amount.

M-Powder
A convenient sachet format for people who want flexibility and portability in daily use.

M-Lollies
A simple oral format for people who want the easiest daily option to continue.
Questions people often want answered before they buy
What is the recommended daily use?
Is MAF Capsules Triple a medicine?
Where is it made and where does it ship from?
Do you provide medical treatment in Australia?
What should I know about shipping, cancellations and returns?
Featured Research
A selection of key studies from our research library.
Telomere Length in Young and Aged Mice
Study on increased telomere length in mice.
MAF and Cognitive Health in Elderly
Impact of MAF on cognitive performance in seniors.
Degalactosylated Whey Protein & Inflammation
Suppression of inflammatory responses in mice.
MAF Capsules in Hospitalized Patients
Clinical outcomes in non-critical COVID-19 cases.
Educational Articles
A selection of featured articles on longevity, immune support, and healthy aging.
Exploring Links Between Nutrition and Longevity
A feature article exploring research on nutrition, immune support, telomeres, Klotho, and healthy aging.
Could Immune Proteins Have Anti-ageing Benefits?
An article examining whether immune-related proteins may play a role in supporting healthy aging pathways.
How Can You Protect Your Cells From Aging?
A science article focused on cellular aging, resilience, and strategies related to protecting long-term cell health.
Begin with Saisei’s flagship format
Choose MAF Capsules Triple for a premium, research-led daily routine designed to support healthy aging, immune balance and cellular vitality.













